
The central atom, Be, has two pairs of bonding electrons. 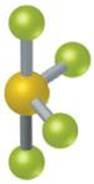
This is a molecule with fewer than 8 valence electrons. What is the Lewis structure of BeCl 2 ? Be has 2 valence electrons, and each ClĬontributes 7 valence electrons, for a total of 16 electrons. With 90 obetween all of the valence electrons. The groupsĪbove and below the central triangle are at a 90 o angle to the triangle.Įlectrons separated in space will form an octahedron, This shape consists of a triangle with two electron pairsĪngles of the middle, triangular portion are 120 o. The bond angle in a tetrahedron is 109.5 o. The bond angle in this structure is 120 o.Īre separated as far as possible from each other. The angle between the lone pairs and the central atom is 180 o.Įlectrons pairs get as far apart from each other, a trigonal planar structure is formed, as shown below. The pairs of dots ( :) represent two pairs of valenceĮlectrons forming bonds to the central atom. Try to get as far away from each other as possible, a linear shape is formed. These electron pairs will keep as far away from each other, due to The concept is that valence shell electron pairs are involved in bonding, and that Lewis structures can give us an approximateĪ simple procedure that allows us to predict overall geometry is the VSEPR, Valence Shell Electron Pair Repulsion. doi: 10.1021/ed083p336.2.The bond lengths and angles, are determined experimentally. "Mechanisms that Interchange Axial and Equatorial Atoms in Fluxional processes: Illustration of the Berry Pseudorotation, the Turnstile and the Lever Mechanisms via animation of transition state normal vibrational modes". "Dynamic nuclear magnetic resonance study of fluorine exchange in liquid sulfur tetrafluoride". "The Trigonal Bipyramid Geometry (SN = 5) (VSEPR Part 4)" (PDF). The following compounds and ions have disphenoidal geometry: Sulfur tetrafluoride is the premier example of a molecule with the disphenoidal molecular geometry (see image at upper right). The four atoms in motion act as a lever about the central atom for example, the four fluorine atoms of sulfur tetrafluoride rotate around the sulfur atom. Thus, the 19F NMR spectrum of SF 4 (like that of PF 5) consists of single resonance near room temperature. This exchange of positions results in similar time-averaged environments for the two types of ligands. The ideal angle between the axial ligands and the equatorial ligands is 90° whereas the ideal angle between the two equatorial ligands themselves is 120°.ĭisphenoidal molecules, like trigonal bipyramidal ones, are subject to Berry pseudorotation in which the axial ligands move to equatorial positions and vice versa. Typically the bond distance to the axial ligands is longer than to the equatorial ligands. The equatorial pair of ligands is situated in a plane orthogonal to the axis of the axial pair. The axial pair lie along a common bond axis so that are related by a bond angle of 180°. Structure Ĭompounds with disphenoidal (see-saw) geometry have two types of ligands: axial and equatorial. Repulsion by bonding pairs at 120° is much smaller and less important. An equatorial lone pair is repelled by only two bonding pairs at 90°, whereas a hypothetical axial lone pair would be repelled by three bonding pairs at 90° which would make it stable. This is true because the lone pair occupies more space near the central atom (A) than does a bonding pair of electrons. An atom bonded to 5 other atoms (and no lone pairs) forms a trigonal bipyramid with two axial and three equatorial positions, but in the seesaw geometry one of the atoms is replaced by a lone pair of electrons, which is always in an equatorial position. The seesaw geometry occurs when a molecule has a steric number of 5, with the central atom being bonded to 4 other atoms and 1 lone pair (AX 4E 1 in AXE notation). Most commonly, four bonds to a central atom result in tetrahedral or, less commonly, square planar geometry. The name "seesaw" comes from the observation that it looks like a playground seesaw. Structural molecular geometry Seesaw molecular geometryĭisphenoidal or seesaw (also known as sawhorse ) is a type of molecular geometry where there are four bonds to a central atom with overall C 2v molecular symmetry.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
